Cosmos Health Introduces the "18 Series " in the United States - A Science-Driven Nutraceutical Platform Built on Patented Ingredients, Published Clinical Research, and Exact Clinical Dosing; Targeting a Portfolio of 18 Clinically Validated Products
Science-first nutraceutical platform: Every "18 " product must clear three criteria before reaching market - a patented ingredient, peer-reviewed clinical evidence, and the exact dose studied in clinical trials; a bar the vast majority of the supplement industry cannot meet
Three products introduced, $7.8 billion addressable market: Noor18™, Liv18™, and Cur18™ - targeting healthy aging & beauty, liver & metabolic support, and inflammation & mobility across three of the fastest-growing health categories in the U.S.
Institutional-grade scientific standard: Patented ingredients, published randomized controlled trials, exact clinical dosing - built to the standard of institutional science, not marketing
Compounding platform value, 18 clinically validated products targeted: Each new "18 " product retroactively strengthens every existing one - the portfolio becomes more valuable with every addition
CHICAGO, IL / ACCESS Newswire / April 21, 2026 /Cosmos Health Inc. ( "Cosmos Health " or the "Company ") (NASDAQ:COSM), a diversified, vertically integrated global healthcare group, today announced the U.S. introduction of the "18 Series " - a premium nutraceutical platform designed to meet growing consumer demand for clinically validated health solutions.
1. The 18 Series - Overview
Cosmos Health Inc. is executing a disciplined, science-first strategy to build the what we believe to be most credible clinical supplement portfolio in the United States. The cornerstone of this strategy is the '18 Series ' - a portfolio of products where every brand carries a shared scientific promise: patented ingredient, published clinical evidence, exact clinically studied dose.
The '18 Series ' is not a product line in the traditional sense. It is a certification - a quality threshold that each product must clear before it earns the right to carry the designation. This creates a compounding brand equity effect: each new '18 ' introduction strengthens the credibility of every existing '18 ' product, and the portfolio as a whole becomes more valuable than the sum of its parts.
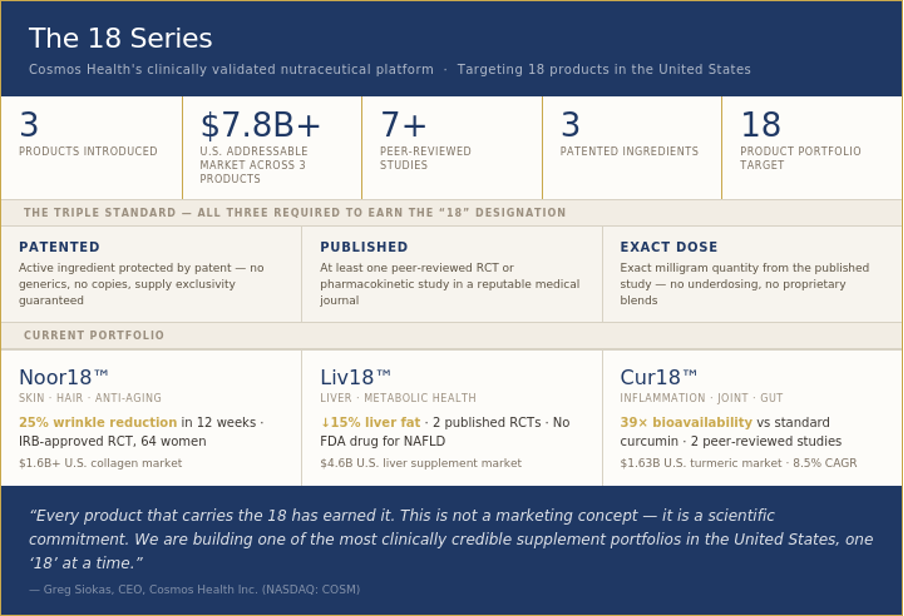
Three products are currently in the '18 Series ': Noor18™ (marine collagen, anti-aging), Liv18™ (liver fat support), and Cur18™ (next-generation curcumin). Together, they address a combined U.S. supplement market opportunity exceeding $7.8 billion, across three of the fastest-growing health categories in the country - with a long-term strategic target of 18 products spanning key biological systems.
18 Series Products | Combined Market | Clinical Studies | Patents | Portfolio Target |
3 introduced | $7.8B+ U.S. addressable market across current categories | 7+ peer-reviewed published studies | 3 patented active ingredients | 18 products targeted across key biological systems |
2. The "18 " - A Scientific Standard, Not a Marketing Claim
The choice of '18 ' as the unifying symbol of the Company 's premium portfolio is deliberate and multi-layered. It is simultaneously a scientific reference, a portfolio classification system, and a philosophical statement about what this company believes supplements should be.
2.1 The Scientific Origin
The '18 ' traces its origin to a landmark pharmacokinetic study - an 18-subject, randomized, double-blind crossover trial that demonstrated substantially enhanced bioavailability of curcumin versus the standard extract. That number became a symbol: the moment science proved that the supplement market had been under-delivering on its biggest promises.
2.2 The Portfolio Classification
Within the Cosmos Health brand architecture, the '18 ' suffix is a certification mark. A product earns the '18 ' designation only when it satisfies all three criteria simultaneously:
Patented:The active ingredient is protected by an active patent covering composition, manufacturing process, and/or health application - preventing generic competition and ensuring supply exclusivity
Published:At least one peer-reviewed, published randomized controlled trial or pharmacokinetic study in a reputable medical journal substantiates the primary health claim at the commercial dose
Exact dose:The product delivers the same milligram quantity used in the published study - no underdosing, no proprietary blends that obscure ingredient amounts
2.3 The Philosophy - Chai (חַי)
In Hebrew tradition, the number 18 represents Chai - meaning 'life. ' It is one of the most universally recognized symbols of vitality, protection, and renewal across cultures. Cosmos Health does not use this symbolism in a religious context. Rather, the '18 ' embodies the brand 's core mission: every product in the series is designed to protect and renew a critical biological system - liver health, collagen architecture, cellular inflammation response. The '18 ' is life, made scientifically rigorous.
3. The 18 Series - Product by Product
Noor18™ - Marine Collagen · Anti-Aging · Beauty from Within
Noor18™ is powered by a patented marine collagen tripeptide ingredient derived from wild-caught fish using patented enzymatic hydrolysis technology. The product targets the single largest consumer driver in the premium supplement market: visible anti-aging results - reduced wrinkles, thicker hair, stronger nails, improved joint comfort.
Noor18™ enters the $1.6 billion U.S. collagen supplement market as the premium clinical alternative, directly benchmarked against leading DTC collagen brands.
Noor18™ Profile | Detail |
Clinical Evidence | IRB-approved RCT: 64 women, 12 weeks, 25% wrinkle reduction |
Key Claims | Reduces wrinkles · Improves skin hydration & elasticity · Thickens hair · Strengthens nails · Joint support |
Market | $1.6B+ U.S. collagen supplement market · Women 35-65 primary demographic |
Liv18™ - Liver Fat Support · Metabolic Health · NAFLD/MASLD
Liv18™ is powered by a patented botanical blend clinically demonstrated to reduce liver fat accumulation in two independent randomized controlled trials. It enters the U.S. market at a critical moment: NAFLD/MASLD now affects more than 30% of U.S. adults, there is no FDA-approved drug for the condition, and consumer awareness of liver health is rising sharply.
Liv18™ is the only consumer supplement in the U.S. market delivering this patented botanical blend at the exact clinical dose - the pure, single-ingredient, clinically dosed version.
Liv18™ Profile | Detail |
Clinical Evidence | 2 published RCTs: Study 1 (Type 2 Diabetes, 14 weeks) + Study 2 (Non-diabetics >50, 12 weeks) |
Key Claims | ↓15% liver fat (>50yr subjects) · ↓ALT/AST enzymes · 2× weight loss vs placebo |
Competitive Moat | Only consumer supplement at the exact clinical dose in the U.S. market · No FDA drug for NAFLD - no Rx competition |
Market | $4.6B U.S. liver health supplement market · 7% CAGR through 2030 |
Cur18™ - Next-Gen Curcumin Therapy · Inflammation · Joint · Gut · Mood
Cur18™ is powered by a patented curcumin extract, with GRAS self-affirmation underway, supported by published pharmacokinetic and clinical efficacy data. It enters one of the largest and most consumer-familiar supplement categories in the United States: the $1.63 billion turmeric/curcumin market - with a product that is categorically better than anything on shelf.
The curcumin category is vast but deeply commoditized. The overwhelming majority of turmeric supplements use standard extracts with known absorption limitations.Cur18™ is the disruptive alternative: same botanical source, dramatically enhanced bioavailable payload, GRAS self-affirmation underway, patented technology, and two peer-reviewed studies. The bioavailability story is one of the most compelling science-to-consumer narratives in the supplement industry.
Cur18™ Profile | Detail |
Bioavailability | 39× enhanced free curcumin bioavailability vs standard curcumin extract |
Clinical Evidence | Pharmacokinetic study (Medicine, 2021, n=18) + Osteoarthritis RCT (Nutrients, 2021, n=101) |
GRAS Status | GRAS self-affirmation process completed by an independent expert panel, pending final confirmation |
Key Claims | Anti-inflammatory · Joint pain · Gut health · Mood · Post-exercise recovery · Antioxidant |
Competitive Moat | 39× bioavailability - the most powerful efficacy claim in the curcumin category |
Market | $1.63B turmeric supplement market (2025) → $3.12B by 2033 · 8.5% CAGR |
4. Brand Architecture - How the Portfolio Compounds
The '18 Series ' is architected for compounding brand value. Each product introduction does not merely add a revenue line - it strengthens the entire portfolio 's credibility. This is the fundamental difference between a product line and a platform.
4.1 Shared Visual Identity
Every '18 ' product shares the same visual language: Deep Navy × Prestige Gold × Parchment - premium, clinical, and unmistakably consistent. On shelf and online, a consumer who knows Noor18™ immediately recognizes Liv18™ and Cur18™ as members of the same trusted family.
4.2 Cross-Product Consumer Journey
The target consumer for the '18 Series ' is the health-conscious adult aged 35-65 who is willing to pay a premium for supplements that work - and who is frustrated by the gap between what the supplement industry promises and what it delivers. This consumer is the same person across all three products: they want better skin AND better liver health AND better inflammation control. The '18 Series ' is designed to be a complete premium health stack - not three separate purchase decisions, but one brand relationship.
4.3 The Compounding Effect
Each new '18 ' introduction creates retroactive value for existing products. When Cur18™ was introduced, it told the world that the company that introduced Liv18™ has now applied the same standard to curcumin. When the next '18 ' is introduced, it will tell the world that the standard continues to hold. The brand is intended to be self-reinforcing. Investors, retailers, and consumers will learn to trust the '18 ' before they know what it contains.
Platform Driver | Effect |
Unified brand identity | Premium, consistent visual language across all products reinforces scientific credibility at shelf and online |
Cross-category reach | Each new product opens a new high-growth health category under the same trusted standard |
Compounding credibility | Every new '18 ' introduction retroactively strengthens existing products - the portfolio appreciates with each addition |
Consumer stack opportunity | The same 35-65 health-conscious consumer is the target across all three products - one relationship, multiple revenue streams |
Greg Siokas, CEO of Cosmos Health, stated: "We chose 18 because it earns its meaning twice. Once in the laboratory, where science changed what we know about absorption. And once in the deepest tradition of human wellness, where 18 has always meant life. When consumers see the '18 ', they should know: this product has been held to the highest standard we know how to set.
I started this company with one conviction: that the health supplement industry was systematically under-delivering on its promises, and that the only way to change that was to build products on a foundation of real science. Not marketing claims. Not proprietary blends designed to hide underdosing. Real peer-reviewed, published, patented science. The '18 Series ' is the embodiment of that conviction. Every product we have introduced under the '18 ' designation - Noor18™, Liv18™, Cur18™ - has met a standard that the vast majority of the supplement industry cannot match. A published RCT. A patented ingredient. The exact dose used in the study. That is not the norm in this industry. It should be. And we are going to make it the norm, one product at a time. For our shareholders, I want to be direct: we are not building a supplement company. We are building a clinical evidence platform, dressed as a consumer brand. The '18 ' is our moat. Every new product that earns the designation makes the ones before it more valuable. The addressable market is enormous - $7.8 billion across just three categories. We have only begun. The pipeline is deep, the standard will never drop. The 18 Series is how Cosmos Health becomes a household name - not by selling more, but by being trusted more. "
About Cosmos Health Inc.
Cosmos Health Inc. (Nasdaq:COSM), incorporated in 2009 in Nevada, is a diversified, vertically integrated global healthcare group. The Company owns a portfolio of proprietary pharmaceutical and nutraceutical brands, including Sky Premium Life®, Mediterranation®, bio-bebe®, C-Sept® and C-Scrub®. Through its subsidiary Cana Laboratories S.A., licensed under European Good Manufacturing Practices (GMP) and certified by the European Medicines Agency (EMA), it manufactures pharmaceuticals, food supplements, cosmetics, biocides, and medical devices within the European Union. Cosmos Health also distributes a broad line of pharmaceuticals and parapharmaceuticals, including branded generics and OTC medications, to retail pharmacies and wholesale distributors through its subsidiaries in Greece and the UK. Furthermore, the Company has established R&D partnerships targeting major health disorders such as obesity, diabetes, and cancer, enhanced by artificial intelligence drug repurposing technologies, and focuses on the R&D of novel patented nutraceuticals, specialized root extracts, proprietary complex generics, and innovative OTC products. Cosmos Health has also entered the telehealth space through the acquisition of ZipDoctor, Inc., based in Texas, USA. With a global distribution platform, the Company is currently expanding throughout Europe, Asia, and North America, and has offices and distribution centers in Thessaloniki and Athens, Greece, and in Harlow, UK. More information is available at www.cosmoshealthinc.com, www.skypremiumlife.com, www.cana.gr, www.zipdoctor.co, www.cloudscreen.gr, as well as LinkedIn and X.
Forward-Looking Statements
With the exception of the historical information contained in this news release, the matters described herein may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Words such as "believes, " "expects, " "anticipates, " "intends, " "projects, " "estimates, " "plans, " and similar expressions, or future or conditional verbs such as "will, " "should, " "would, " "may, " and "could, " generally identify forward-looking statements, although not all forward-looking statements contain these words. These statements involve risks and uncertainties that may individually or materially affect the matters discussed herein for a variety of reasons outside the Company 's control, including, but not limited to: the Company 's ability to raise sufficient financing to implement its business plan; the effectiveness of its digital asset strategies, including accumulation and yield-generating activities; the impact of the war in Ukraine and ongoing conflicts in the Middle East and other regions on the Company 's business, operations, and the economy in general; and the Company 's ability to successfully develop and commercialize its proprietary products and technologies. Readers are cautioned not to place undue reliance on these forward-looking statements, as actual results could differ materially from those anticipated. Readers are encouraged to review the risk factors set forth in the Company 's filings with the SEC, which are available at the SEC 's website (www.sec.gov). The Company disclaims any obligation to update or revise forward-looking statements, whether as a result of any new information, future events, or otherwise.
Investor Relations Contact:
BDG Communications
cosm@bdgcommunications.com
SOURCE: Cosmos Health Inc.
View the original press release on ACCESS Newswire
© 2026 ACCESS Newswire. All Rights Reserved.



















